 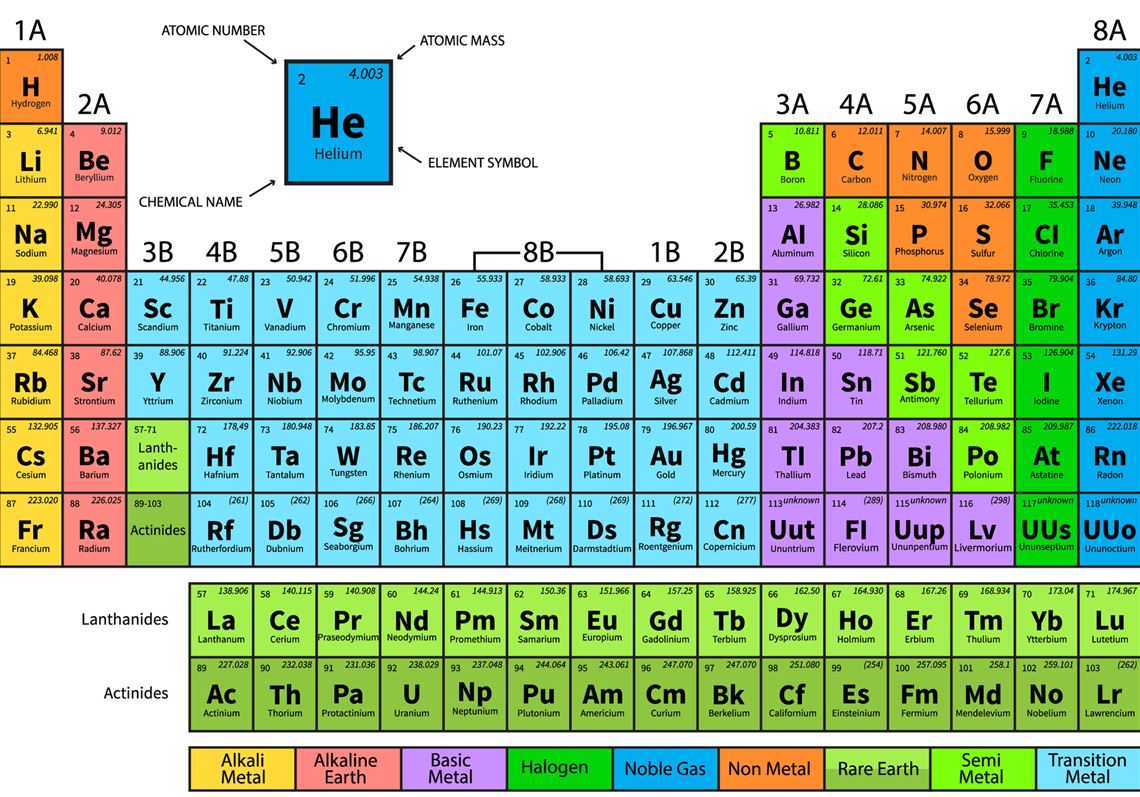
Because its 2n shell is filled, it is energetically stable as a single atom and will rarely form chemical bonds with other atoms. The periodic table of elements is an arrangement of the chemical elements ordered by atomic number in columns (groups) and rows (periods) presented so as to. The Periodic Table of Chemical Elements is one of. For instance, lithium ( Li \text Ne start text, N, e, end text ), on the other hand, has a total of ten electrons: two are in its innermost 1 s 1s 1 s 1, s orbital and eight fill the second shell-two each in the 2 s 2s 2 s 2, s and three p p p p orbitals, 1 s 2 1s^ 2 1 s 2 1, s, squared 2 s 2 2s^ 2 2 s 2 2, s, squared 2 p 6 2p^6 2 p 6 2, p, start superscript, 6, end superscript. A yearlong initiative to raise awareness of chemistry and its applications for sustainable development. Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. Using the Periodic Table of the Elements, the properties of elements can often be used to deduce the properties of neighboring elements. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. (credit a: modification of work by Serge Lachinov credit b: modification of work by Den fjättrade ankan/Wikimedia Commons) Video 3.2.2: An introduction to the organization of the periodic table into periods and groups. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Figure 3.2.1: (a) Dimitri Mendeleev is widely credited with creating (b) the first periodic table of the elements. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. 1 2 Period 2 has much more conclusive trends. 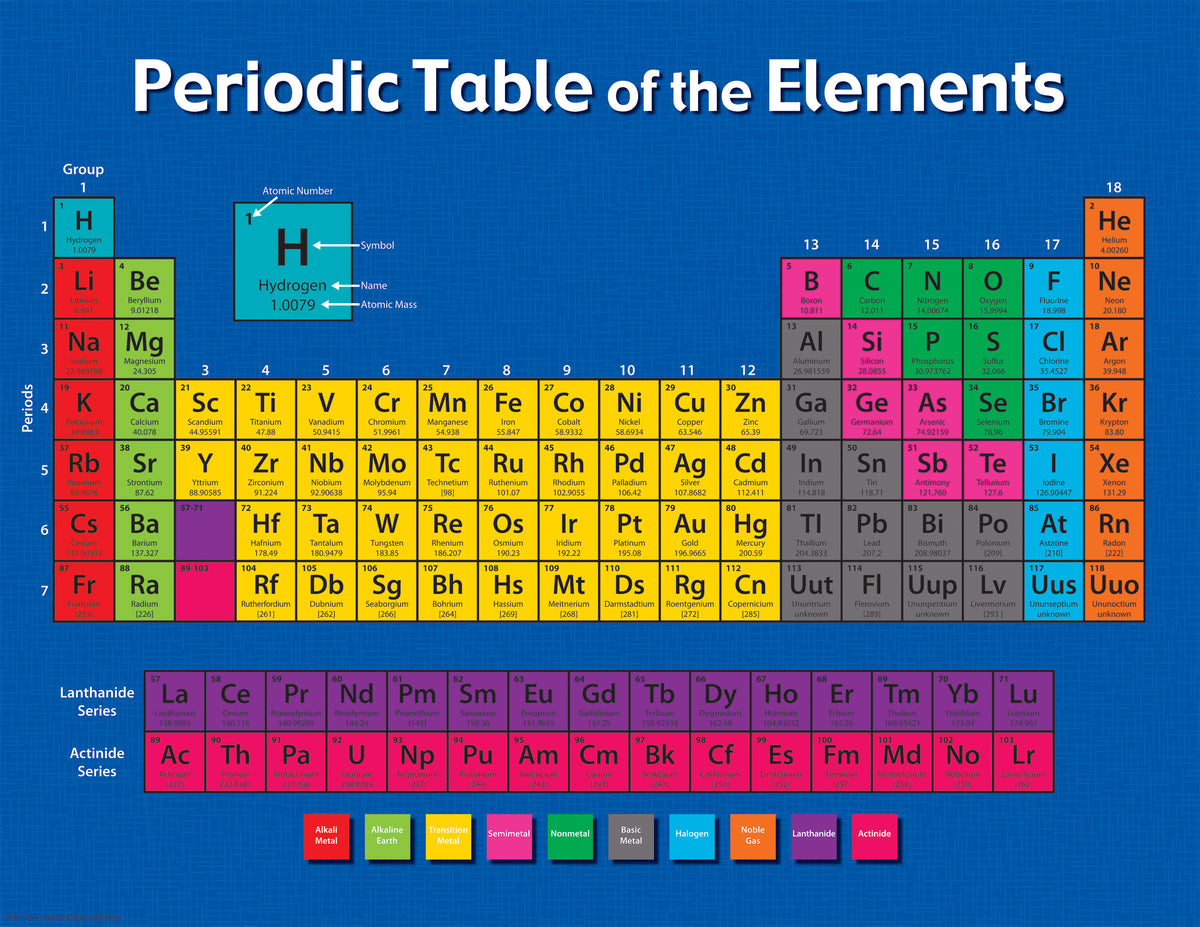
Period 1, which only contains two elements ( hydrogen and helium ), is too small to draw any conclusive trends from it, especially because the two elements behave nothing like other s-block elements. The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital. Period 2 is the first period in the periodic table from which periodic trends can be drawn. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
